| References |
| Synonyms |
- Peroxisome Proliferator-activated Receptor γ Ligand Binding Domain
|
| Formulation |
A solution in 50 mM sodium phosphate, pH 7.2, containing 20% glycerol, 150 mM sodium chloride, and 1 mM DTT |
| Purity |
≥90% |
| Stability |
6 months |
| Storage |
-80°C |
| Shipping |
Dry ice
in continental US; may vary elsewhere
|
Background Reading
Kersten, S., Desvergne, B., and Wahli, W. Roles of PPARs in health and disease. Nature 405 421-424 (2000).
Rosen, E.D., and Spiegelman, B.M. PPAR.gamma: A nuclear regulator of metabolism differentiation, and cell growth. J Biol Chem 276(1) 37731-37734 (2001).
Mudaliar, S., and Henry, R.R. New oral therapies for type 2 diabetes mellitus: The glitazones or insulin sensitizers. Annu Rev Med 52 239-257 (2001).
Uppenberg, J., Svensson, C., Jaki, M., et al. Crystal structure of the ligand binding domain of the human nuclear receptor PPARγ. J Biol Chem 273 31108-31112 (1998).
Show all 4
Hide all but first 3
| Size |
Global Purchasing |
| 25 µg |
|
| 50 µg |
|
| 100 µg |
|
Description
Source:
recombinant N-terminal hexahistidine-tagged protein expressed in E. coli
·
Mr:
~34 kDa
·
Protein concentration:
0.30 mg/ml
·
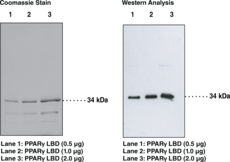
The peroxisome proliferator activated receptor (PPAR) family of ligand-activated transcription factors consists of three subtypes encoded by separate genes: PPARα, PPARδ (also referred to as hNUC1, PPARβ, or FAAR), and PPARγ.1 Among them PPARγ is the most widely studied and has been implicated in the pathology of numerous diseases including obesity, diabetes, atherosclerosis, and cancer.1,2 Structurally, PPARγ can be described by two primary domains - a ligand binding domain (LBD) and a DNA binding domain (DBD).2,3 Binding of activating ligands to the LBD promotes heterodimerization with retinoic acid-like receptor (RXR) resulting in the regulated expression of target genes, a significant number of which are related to lipid metabolism.1 Select members of the thiazolidinedione class of PPARγ ligands, which includes rosiglitazone and pioglitazone, are currently used for the treatment of insulin resistance associated with type II diabetes.4 Cayman’s PPARγ LBD is expressed and purified from E. coli. The purity was determined using gel electrophoresis followed by coomassie staining and western blot analysis using a PPARγ polyclonal antibody. The functionality was tested using a fluorescent ligand binding assay.
1
Kersten, S., Desvergne, B., and Wahli, W. Roles of PPARs in health and disease. Nature 405 421-424 (2000).
2
Rosen, E.D., and Spiegelman, B.M. PPAR.gamma: A nuclear regulator of metabolism differentiation, and cell growth. J Biol Chem 276(1) 37731-37734 (2001).
3
Uppenberg, J., Svensson, C., Jaki, M., et al. Crystal structure of the ligand binding domain of the human nuclear receptor PPARγ. J Biol Chem 273 31108-31112 (1998).
4
Mudaliar, S., and Henry, R.R. New oral therapies for type 2 diabetes mellitus: The glitazones or insulin sensitizers. Annu Rev Med 52 239-257 (2001).
|